Indonesia’s food and drug agency has revoked fever syrup production licenses for two local firms for violating manufacturing rules.
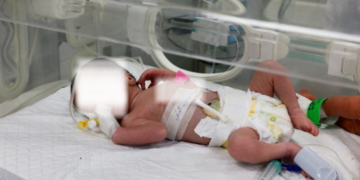
This is in relation to a probe that was launched into the death of 150 children, most under five years, due to acute kidney injury.
The agency suspected the acute kidney injuries were caused by ethylene glycol and diethylene glycol, present in some syrup-based medications.
The two ingredients are used in antifreeze and brake fluids and other industrial applications but also as a cheaper alternative in some pharmaceutical products to glycerine, a solvent or thickening agent in many cough syrups.
The Indonesian food and drug authority (BPOM), banned PT Yarindo Farmatama and PT Universal Pharmaceutical Industries, launching criminal action against them.
BPOM chief Penny K. Lukito, said the two firms produced drugs with substandard raw materials, failed to report a change in ingredients and used some materials in excess of guidelines.
The agency has sworn to investigate the importers of the two alternative ingredients and ensure they were never sold to pharmaceutical industries.
PT Universal Pharmaceutical Industries declined to comment, citing the ongoing investigation.
Indonesia imports its raw ingredients for medicine mostly from China and India.
Also Read: India:100 million Doses of Expired Covid 19 Vaccine Destroyed